 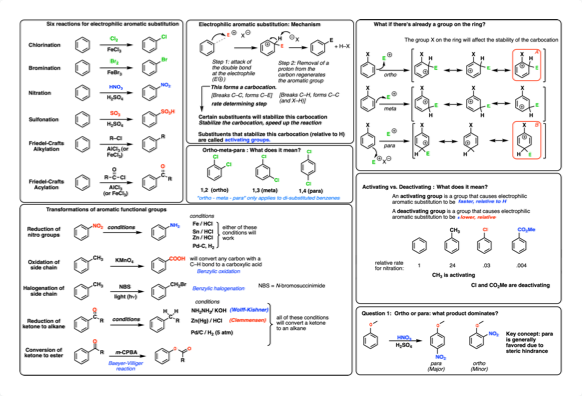
The calculated free activation energy of 23.1 kcal/mol shows that bisphenol A-3,4-quinone can form an adduct with deoxyguanosine and can be a mutagen. To study the possible genotoxic mechanisms of bisphenol A, the chemical reactivity of the metabolite bisphenol А-3,4-quinone with deoxyguanosine was calculated using the density functional theory in combination with the Langevin dipole solvation model. RMS displacement (MSD) of water and phenol for various systems show that TBP can limit the migration of water and phenol near the boundary section. 
The radial distribution function (RDF) of TBP and phenol shows that most of the hydrogen bond interaction distance is 1.8 Å. Systems of the organic phase with different concentrations of tributyl phosphate (TBP) and the water phase with different concentrations of phenol were modeled. Using molecular dynamics modeling by means of the GROMACS software package, interfacial features and the phenols extraction process have already been studied. 19 Moreover, they are the only nonindirect source of information about the structure and energy of transition states, which, in principle, cannot be experimentally observed. Quantum chemical calculations are also used to describe reaction mechanisms. 
However, the correctness of the calculations should be confirmed by the correspondence of the obtained results with the experimental physical parameters. In difficult cases, the use of quantum chemical calculations can predict the behavior of molecules in various cases. Quantum chemical calculations are currently used by researchers in various fields of physics and chemistry more and more often. Mayer, in Advances in Quantum Chemistry, 2020 1.2 Theoretical and experimental research on bisphenol A 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
